My goal is to help you understand chemistry by breaking down difficult concepts into pieces you can understand. Need more help? Check out some of my playlists! The number of electrons is a bit more complicated, and must be found through the charge of the atom. You find the number of neutrons in an element by subtracting the atomic number from the atomic number. Examples: 40 18 Ar, 40 20 Ca In the case of Ca and Ar atomic mass of both the element is 40. 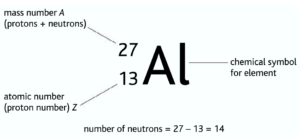
Isobars: These are atoms of the elements having the same atomic mass but different atomic numbers. 'Mass number'-'atomic number''number of neutrons' ''16'O'(2-) has 8 protons, 8 neutrons, and 10 electrons. The number of neutrons in Carbon and Nitrogen is 8. The number of neutrons will be the same as the number of protons: it is also 20. If we know the atomic number (8), we can subtract that from the mass number to determine the number of neutrons. 
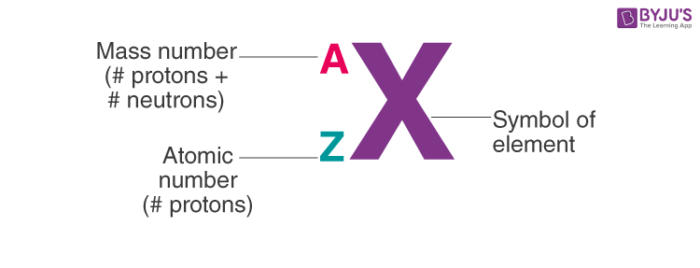
Thus, the three isotopes of hydrogen are represented as follows: Protium: 1 1 H. You just need to know a couple tricks to always be able to find the number of protons neutrons and electrons! The number of protons in an atom is the same as the atomic number. It is also the number written as a superscript to the left of the isotopic symbol (e.g. The isotopes of an element are usually represented by putting the atomic number as the subscript and their mass number as the superscript on the symbol of the element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
